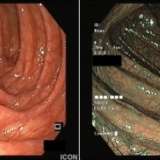
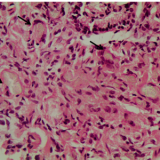
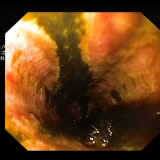
This seemed to have been a particularly difficult case. Although the stricture looks peptic, the wide-spread gastric intestinal metaplasia tells you that the patient has come to the end of a life-time of Helicobacter infection. She now has atrophic gastritis with florid intestinal metaplasia and is of course achlorhydric. Without any gastric acid, how could she possibly have developed a reflux-induced oesophageal stricture !!?
The strictures seen in eosinophilic oesophagitis are in my experience less “membrane-like” and 2-3 cm in length. Furthermore, there are no oesophageal stigmata of the condition (longitudinal furrows, micro-abscesses or trachelisation). Option D (intermittent symptoms) implies oesophageal incoordination. However, if this was the underlying condition, she should not have a stricture. Finally this does not look like a squamous cell carcinoma (option e).
This leaves us with drug induced stricturing (NSAID or biphosphonate). Indeed the patient had recently been started on alendronate! Well done Dr Lekharaju who got the correct diagnosis!
In addition to stricturing, biphosphonate may cause an acute oesophagitis, oesophageal ulceration and even gastric ulcers, presumably due to direct contact of the tablets with the mucosa. It seems that alendronate is particularly hazardous whilst risedronate (a third generation biphosphonate), may be less ulcerogenic. The co-administration of a NSAID and alendronate is especially toxic.
Classically, the treatment includes stopping the biphosphonate and starting a proton pump inhibitor. However, it must be tempting to restart after a couple of months with a lower dose of some other biphosphonate, advice the patient to take the tablets and then eat some food (although you are supposed to take alendronate on an empty stomach) and continue the proton pump inhibitor. Not sure what risk to quote the patient for a relapse. However, if the patient has marked osteoporosis, it may be a risk worth taking?

Please log in with your myUEG account to post comments.