Correct answer: d.
Discussion
Hematoxylin has a deep blue-purple colour and stains nucleic acids, while eosin is pink and stains proteins in a nonspecific manner. Liver specimens stained with H&E will, therefore, typically display blue nuclei, with the cytoplasm and extracellular matrix in different shades of pink. Masson’s trichrome stain is commonly used in parallel with the H&E stain to examine liver specimens. With this method, the three dyes are used selectively to stain collagen (in blue), muscles, erythrocytes, fibrin and cytoplasm (in red), with nuclei often stained black.
For both stains, control mice (figures 1 and 2) show normal liver architecture and cellular integrity with no fibrosis. Of note, because Masson’s trichrome stains type 1 collagen, portal tracts and vessel walls will also be stained blue, although not necessarily representing reactive fibrosis (figure 2).
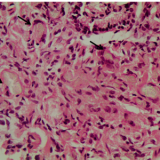
Hepatocellular steatosis can be identified by either the presence of single large fat droplets, alongside nuclei dislocation to the cell’s periphery (macrovesicular steatosis), or small lipid droplets and no nuclei displacement (microvesicular steatosis). The former is typical of NAFLD and associates with inflammation in NASH (figure 3). Steatosis may also be observed concomitantly with fibrosis, whose characteristic pattern in NASH is perisinusoidal/pericellular (‘chicken wire’) (figure 4). In more advanced stages of fibrosis, collagen fibres become thicker and form a more widespread network (figure 5). While cirrhosis appears to be absent (inconclusive evidence of extensive scar tissue, inflammatory infiltrates and the presence of regenerative micronodules), it should not be excluded; alternative staining and/or capturing additional images at 100x magnification would be needed to reach a definite conclusion.
During the course of NASH–HCC, typical hepatocellular cytoplasmic (figure 6) and nuclear (figure 7) alterations occur, including cytoplasmic accumulation of protein droplets, lipofuscin, ceroid, copper and iron. Furthermore, in HCC, liver architecture and cellular integrity is severely compromised (figure 8). Here we can note the presence of broad trabeculae and irregular nuclear contours.
Lipofuscin or ceroid is a yellow-brown product considered to represent a lipid breakdown product of cell membranes that accumulates in Kupffer cells and macrophages as a result of hepatocyte necrosis and/or apoptosis. Foci of ceroid-laden macrophages are common in liver fibrosis (figure 9), although their pathological significance remains poorly explored.3 As for protein accumulation, the most common intracellular inclusions comprise Mallory–Denk bodies (MDB; mainly composed of keratin (K) 8, K18, p62 and ubiquitin) and intracellular hyaline bodies (IHB; composed of p62 and/or ubiquitin). A recent study has shown that HCC patients who have liver IHB inclusions have a poor prognosis.4



Please log in with your myUEG account to post comments.