Correct answer: c.
Discussion
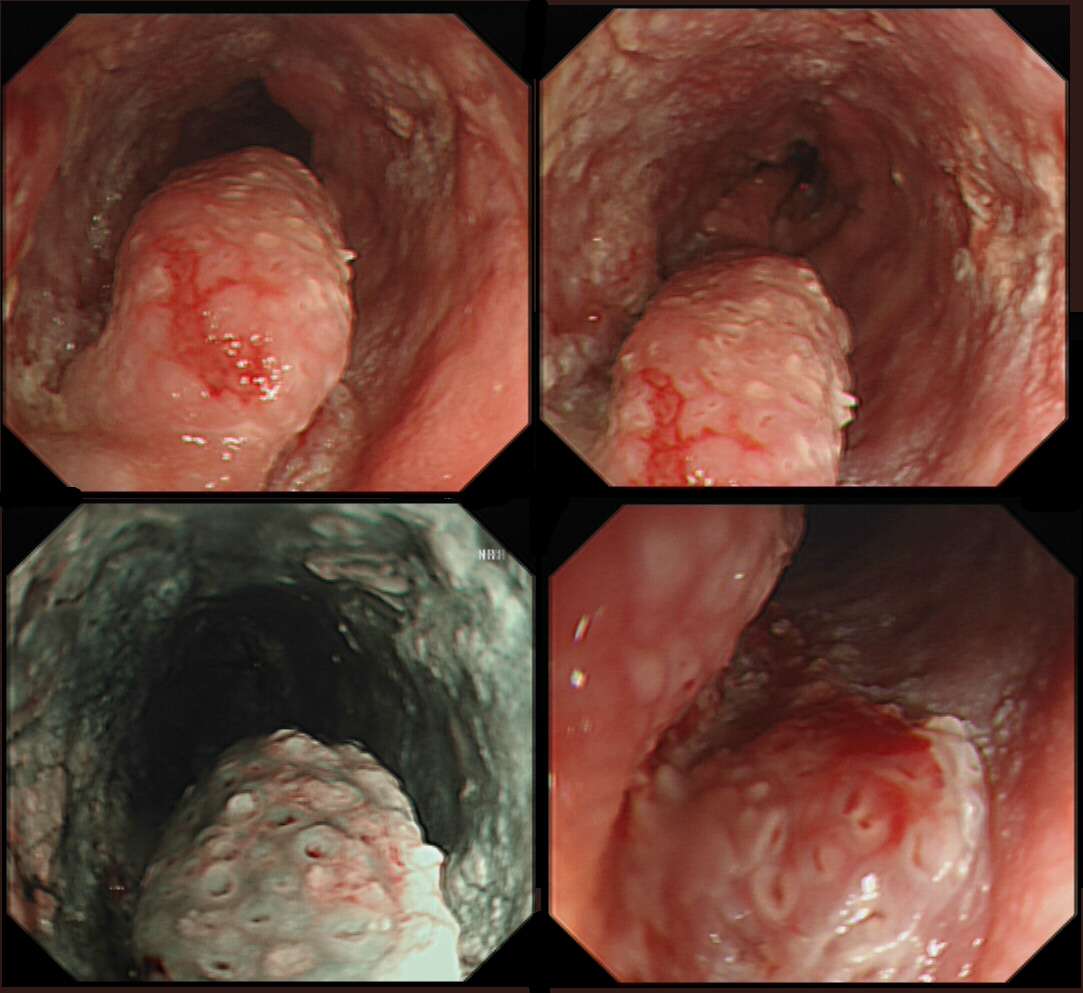
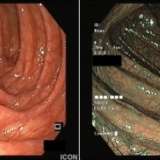
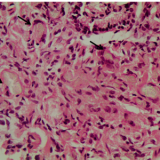
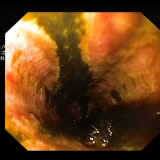
The hyperkeratotic soles visible in the photograph make a diagnosis of tylosis relatively straightforward. However, the difficulty is in deciding the endoscopic nature of the oesophageal nodule. The lesion was removed by endoscopic mucosal resection (EMR) and, in view of the regularly spaced pits, a squamous cell carcinoma seem unlikely. Conversely, the nodule was too large for simple hyperkeratosis. An unrelated diagnosis such as a fibro-epithelial polyp was never likely as clearly the finding was related to Tylosis. In the end, the assumption was that it was a ‘dysplastic, proliferative lesion’. Subsequent histology findings put it in another way—as a ‘squamo-proliferative lesion with moderate dysplasia’.
Tylosis, or keratosis palmaris et plantaris, is a rare autosomal-dominant genetic disorder that is characterised by focal thickening of the palms of the hands and soles of the feet, and appears as cutaneous yellowish thickened plaques.1 There are two distinct classes of tylosis. Type B tylosis usually appears in children by the age of 7 years, but can appear as late as puberty, and is considered to be a benign disease. By contrast, type A (or late-onset) tylosis is associated with a high incidence of squamous cell oesophageal cancer.2
A link between tylosis and oesophageal cancer was first recognised in the 1950s in two large families living in Liverpool, UK.3 These families were revisited in 1994, at which point 89 of the 345 family members had been diagnosed with tylosis, of whom 21 had died of oesophageal cancer.4
Missense mutations in the RHBDF2 gene, which encodes the inactive rhomboid protease RHBDF2, are now known to be the underlying cause of tylosis oesophageal cancer (TOC).5 The estimated lifetime risk of oesophageal squamous cell carcinoma in patients with type A tylosis is thought to be >40% for patients of American ancestry,6 but in excess of 90% in those of British ancestry.3,4 As a consequence of this exceptionally high risk, intensive screening for oesophageal cancer is recommended for patients with type A tylosis. Guidelines suggest that surveillance endoscopy should occur every 1 to 3 years from the age of 30 years, or as soon as the condition is first recognised.7
One study has attempted to characterise the endoscopic appearance of tylosis into mild, moderate and severe disease using both white light endoscopy (WLE) and narrow-band imaging (NBI).8 In this case series of 19 patients, mild tylosis of the oesophagus seen on WLE was described as mild and patchy hyperkeratosis, with intervening normal mucosa. A normal intrapapillary capillary loop (IPCL) pattern was seen under NBI. Severe tylosis was described as confluent hyperkeratotic plaques seen on WLE, and a severely distorted IPCL pattern seen under NBI. The 17 patients described as having mild or moderate tylosis based on the typical endoscopic findings showed no progression of the degree of dysplasia over a mean follow-up period of 30 months, whereas the 2 patients who were classified as having severe tylosis progressed to severe dysplasia, and ultimately underwent an oesophagectomy, following which early squamous cell carcinoma was confirmed in both resected specimens.8


Please log in with your myUEG account to post comments.