Correct answer: e.
Discussion
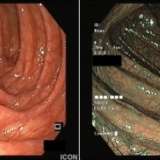
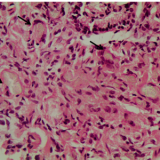
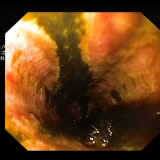
This patient was referred for endoscopic resection; however, the indurated area at 3 o’clock without a discernible crypt pattern concerned me (bottom left image). A subsequent colectomy revealed a tubular adenoma with a patch of high-grade dysplasia (HGD) in the suspicious location. I believe that a Japanese histopathologist would have diagnosed intramucosal cancer.
The topic of dysplasia in colitis has been a roller-coaster ride since Crohn and Rosenberg highlighted a case of rectal cancer complicating ulcerative colitis in 1925.1 Some 25 years later the first description of colon cancer arising in a patient with Crohn’s colitis followed.2 Since then it has become recognised that cancers that develop in the colitic colon are more likely to be small, aggressive and multifocal.3–8
In 1967, Morson and Pang demonstrated that ‘flat dysplasia’ (dysplasia without a visible lesion) is often widespread in colectomy specimens.9 Soon afterwards colonoscopy was making inroads into the teaching hospitals of Europe. The idea that dysplasia seen in random biopsies could identify patients with cancer or those who are likely to develop the disease was attractive.
However, as colonoscopy became established it was soon realised that visible lesions may more directly predict the presence of cancer. In 1981, Blackstone et al.10 coined the term DALM to describe a “Dysplasia-Associated Lesion or Mass”. In their 4-year follow up of 112 patients with colitis, 12 patients developed a DALM and 7 of the 12 were found to also have a synchronous carcinoma.
That mucosal lumps developing in the colitic mucosa were an adverse finding was soon confirmed by others. Rosenstock et al.11 reported on the surveillance of 248 ulcerative colitis patients. Of the 7 patients who had cancer, 5 also had a DALM. Lennard-Jones et al.12 reported on 401 patients and in 5 of their 6 patients who were found to have a cancer, a ‘raised lesion’ was noted on colonoscopy.
However, flat dysplasia also remained a worrying finding. In a blinded study of colectomy specimens, 73% of the specimens with ulcerative colitis and cancer had dysplasia at a site distant from the cancer.13 Similarly, Taylor et al.14 reported that 34% of patients who had colitis and cancer had low-grade dysplasia (LGD) identified elsewhere in their colectomy specimens. These associations were somewhat surprising as it was realised that random biopsy samples only covered some 0.05% of the entire colonic mucosa.11 Clearly the invisible dysplasia must be extensive for random samples to stand any chance of detecting it.
To try to make sense of it all, Bernstein et.al.15 carried out an early meta-analysis of 10 prospective surveillance studies totalling 1,225 patients. Of the 40 patients with a DALM, 17 (43%) were found to have cancer. On the basis of this a general consensus developed that patients with DALM or flat dysplasia should be offered colectomy.
By contrast, there was no consensus about what do to about LGD or when samples had shown ‘indefinite for dysplasia’. On one hand, in Bernstein’s analysis, 210 patients developed LGD at some time during surveillance and of these 17 developed cancer (8.1%).15 Furthermore, 9 of 95 patients (9.5%) who were ‘indefinite for dysplasia’ also developed cancer. On the other hand, the St. Mark’s group published a retrospective analysis showing that about 80% of patients with LGD did not develop either HGD or cancer within 5 years.16
Unfortunately, what appeared to be clear guidelines were difficult to implement in practice.17–20 To begin with, it is almost impossible to distinguish LGD from inflammatory changes.21 In addition, it was clear that the distinction between a DALM and a sporadic polyp was arbitrary as endoscopically and histologically there was no way of distinguishing the two!22,23
People started to consider the option of simply removing the lesions detected by surveillance. After all, not all lesions seemed to carry a high risk of synchronous malignancy.22,24–26 The paradigm that all patients found to have a polyp at surveillance required a colectomy was challenged.27,28
Series of some small prospective case series seemed to support endoscopic resection of lesions. In their review of the surveillance of 332 patients, Connell et al.21 commented that 5 patients who had ‘small polypoid dysplastic lesions’ removed remained well with no evidence of dysplasia or adenocarcinoma 2–13 years later. Similarly, Nugent et al.29 also reported on 5 patients who had a total of 8 pedunculated polyps resected and were then followed up for a median of 6 years (range 3–11 years). All 5 patients were free of cancer at a follow-up of 3–11 years. Medlicott reported on the follow-up of 6 patients who had small polyps removed from outside of the colitic field.25 None developed cancer within a mean follow-up time of 7.2 years.
However, not all reports were reassuring. The St. Mark’s group reported that 3 out of 8 patients who had polyps removed subsequently developed cancer.21 In another 18 patients who had dysplasia detected in their initial biopsy samples, 7 were found to have an unexpected cancer at colectomy.
Rubin published the surveillance findings of 684 patients with ulcerative colitis or Crohn’s disease of at least 8 years’ duration and followed them up for an average of 4.1 years with 103 surveillance colonoscopies.30 At some stage during the study, 48 of the 684 patients had a total of 70 polyps resected and only 1 was malignant. However, the study excluded patients with invisible dysplasia and flat polyps.
Torres retrospectively searched pathology files and found 59 patients with chronic IBD who had a total of 89 small polyps removed (median size 5mm).23 11 patients with flat and sessile lesions were excluded. Follow-up information was only available for 40 patients and after a median follow-up of 13 months, 3 patients had been diagnosed with cancer. The study concluded that the finding of flat dysplasia or flat polyps was of concern.
Bozdech et al.31 reported on the follow-up of 22 patients with ulcerative colitis who had polyps removed. A subsequent cancer developed in 5 patients.
Although the above follow-up studies offered only limited reassurance that it may be relatively safe to remove pedunculated polyps or polyps developing from outside the field of colitis, the argument was put forward that all polyps that could be removed endoscopically should be removed endoscopically, thus saving the patient from a colectomy.32–34
A retrospective review of the St Mark’s surveillance programme, spanning 30 years, highlighted the issue of trying to distinguish DALMs from sporadic adenomas.35 Of the 32 patients diagnosed with ‘indefinite for dysplasia’, 1 patient subsequently developed cancer. The corresponding number of cancers that developed in patients with lesions harbouring LGD was 9 out of 46 and was 7 out of 19 for patients with lesions harbouring HGD. Patients with a DALM had a 31% risk of cancer being discovered in the colectomy specimen compared with 2 cancers developing following an endoscopic resection in 37 patients (5%). The authors admitted that the distinction between a DALM and a sporadic adenoma was arbitrary and that the “…comparison of endoscopically resectable and non-resectable lesions was fraught with bias.” After all, one endoscopist’s unresectable lesion may well be another endoscopist’s resectable lesion.
The issue of invisible flat dysplasia was less contentious. Mayberry et al.36 carried out a meta-analysis in which flat LGD was found to be associated with a ninefold increased risk of subsequently developing cancer. Everyone agrees that surgery should be recommended for patients with endoscopically invisible dysplasia.37–39
On the issue of polyps in colitis, we are now beginning to turn full circle. I was interested to see an abstract from DDW 2015,40 in which the main proponents of the ‘if you can remove – do it!’ camp were beginning to distance themselves from their previous position. The authors concluded that patients with lesions that are large or flat require careful counselling as regards colectomy. You may be surprised to hear that in this study, large was defined as bigger than 10mm!
It is easy to get overwhelmed by the patchy and sometimes contradictory evidence outlined above. To help you navigate these treacherous waters, let me share my own working model of unstable colitis:
The inflamed colitic mucosa is turning over rapidly, which predisposes to the introduction of DNA transcription errors. These errors develop all over the mucosa, but cannot initially been seen by H&E staining or immunohistochemistry (histologically invisible dysplasia). However, as further mutations accumulate our histopathologists begin to see abnormalities (endoscopically invisible, but histologically visible dysplasia). Relatively benign mutations lead to the accumulation of cells in polyps that we can see endoscopically (endoscopically visible flat polyps). More unfortunate mutations lead to complete loss of cellular inhibitions within a stem cell and the growth of small, aggressive cancers.
The above working model fits all the published evidence, but has not been proven. However, an understanding of the published literature and this working model may well help you make the correct decision when confronted with a lesion such as in this case. Of course, you could set out to endoscopically resect the lesion! Naturally, it would be somewhat difficult as the lesion is large and the submucosal lift is always poor in colitis. However, you will now realise that all previous studies would have referred a lesion such as this for surgery! Furthermore, even if you did manage to remove it, the patient may remain at a very high risk of developing an aggressive cancer as some stage in the future. After all the colitic mucosa is likely to be unstable throughout.
Naturally, I do recognise that it is difficult for patients to seriously consider a pan-proctocolectomy and ileo-anal pouch anastomosis when an easily removable flat polyp has developed. Why not just remove it? If you do choose to simply deal with the ‘tip of the iceberg’, make sure that the patient understands that he remains at a greatly elevated risk of cancer and that the intensity of surveillance will have to be increased to at least every 6 months—forever. After all, you and your patient are navigating through an iceberg field and the ship has to slow right down so that when you do hit a submerged iceberg, the damage is limited.


Please log in with your myUEG account to post comments.