Correct answer: e.
Discussion
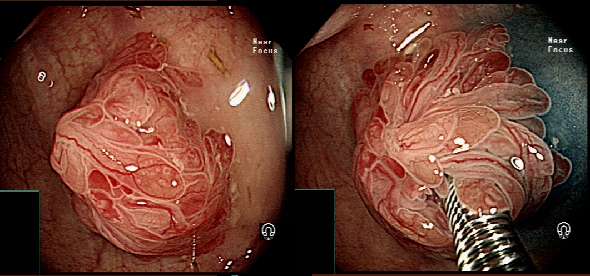
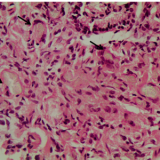
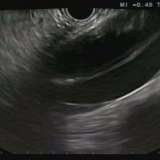
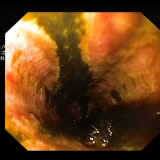
You could be forgiven for guessing that this is a villous adenoma with almost ridiculously long villi. The lesion actually proved to be a traditional serrated adenoma (TSA).
TSAs are part of the complex family of serrated lesions, which currently includes hyperplastic polyps, sessile serrated lesions (SSLs), sessile serrated adenomas (SSAs), TSAs and mixed hyperplastic- adenomatous lesions.
Some 30 years ago, there were only two types of serrated lesion—the hyperplastic polyp and the mixed hyperplastic- adenomatous polyp. In 1990, the TSA was first described by Longare and Fenoglio-Preiser;1 however, there was a period of confusion until Torlakovic et al. reviewed the serrated polyp and gave it its modern name.2 Hyperplastic polyps were not thought to have any malignant potential, but in 1996 Torlakovic and Snover3 reported that the hyperplastic polyps found in hyperplastic polyposis syndrome (now called serrated polyposis syndrome4) were different to ‘normal’ hyperplastic polyps and that they had a malignant potential.
The TSA is a mysterious entity, only accounting for about 1% of colorectal polyps.5 Endoscopically, they can appear as exaggerated tubulovillous adenomas or as villous adenomas. Because of their variable appearance, both endoscopically and histologically, there is no agreement on their molecular features and study findings are conflicting. Hopefully, immunohistochemistry will come to our aid in the future!
It is uncertain how TSAs contribute to cancers arising from the serrated pathway,6 which are thought to give rise to at least the 7–12% of “cancers with a serrated morphology”.7 Serrated carcinomas have a poor prognosis and are more common in the elderly. As more than half of serrated carcinomas arise in the caecum or ascending colon, it is tempting to assume that they originate from hard-to-see serrated precursor lesions. There is some support for this notion because interval cancers are four times more likely than non-interval cancers to be of the CpG island methylator phenotype (CIMP)8 and to show some degree of microsatellite instability, both of which are features of serrated-pathway cancers.9,10 Conversely, it is possible that the one-third of serrated cancers that are found in the distal colon arise from TSAs.11
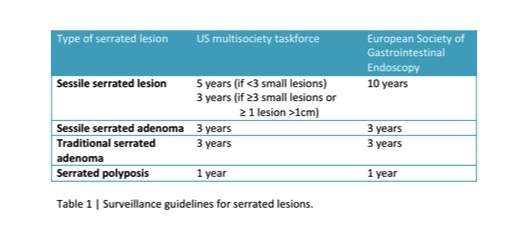
The understanding that some aggressive cancers arise from an alternative pathway to the classic adenoma–carcinoma pathway is a challenge when deciding on surveillance after the removal of a serrated lesion. The US multisociety taskforce on colorectal cancer12 and the European Society of Gastrointestinal Endoscopy13 have both published surveillance guidelines for serrated lesions with some agreement (table 1).

Please log in with your myUEG account to post comments.